Files
Download Full Text (2.4 MB)
Description
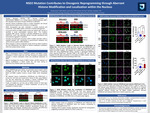
Background: Nuclear Receptor Binding SET Domain Protein 2 (NSD2/WHSC1/MMSET) is a histone methyltransferase, specific to H3K36me2. Commonly, aberrant histone modification levels or localization within the nucleus contribute to dysregulation of gene transcription and cancer development. Most NSD2 mutations, p.E1099K, are identified in its catalytic SET domain, resulting in increased H3K36me2 levels and decreased H3K27me3 levels in acute lymphoblastic leukemia (ALL). However, whether the NSD2 mutation alters histone modification localization within the nucleus remains unclear. This study aims to define NSD2 mutation-driven histone modification and localization in ALL.
Methods: Histone modifications H3K36me2 and H3K27me3 in multiple NSD2 mutant human ALL cell lines and mouse B-cell leukemic cells were determined using Western Blots. The dislocation of H3K36me2 and H3K27me3 in isogenic RCH-ACV cell lines was determined using immunofluorescence via confocal microscopy. Further, isogenic RCH-ACV cell lines with or without NSD2 mutation were treated with a novel NSD2 inhibitor (EX-A5782), followed by Western Blots and immunofluorescence for histone modification level and dislocation detection within the nucleus.
Results: NSD2 mutation drove a significant increase of active marker H3K36me2 and a decrease of repressive marker H3K27me3 in B-ALL, T-ALL cell lines and mouse leukemia cells, leading to oncogenic reprogramming. Surprisingly, the NSD2 mutation caused an aberrant dislocation of H3K36me2, not H3K27me3, to the perinuclear regions. With the treatment of the NSD2 inhibitor, H3K36me2 was reduced in both NSD2 mutant and WT cells, while H3K27me3 was only restored in NSD2 mutant cells. There was no change of H3K27me3 in NSD2 WT cells. NSD2 inhibitor reversed the H3K36me2 distribution in the nucleus as well.
Conclusion: These findings suggest that NSD2 mutation drives an imbalance of H3K36me2 and H3K27me3 and aberrant localization of H3K36me2 within the nucleus, contributing to oncogenic reprogramming in ALL. Targeting NSD2 mutation therapy significantly reversed this dysregulation, suggesting a potential treatment for NSD2 mutant ALL.
Publication Date
2-2-2026
Keywords
NSD2 mutation, H3K36me2, nuclear localization, epigenetic reprogramming, acute lymphoblastic leukemia, epigenetic therapy
Disciplines
Medicine and Health Sciences | Oncology
Recommended Citation
Dasuri, Venkata; Sey, PhD, Samuel; Yuan, Tian; and Li, Jianping, "NSD2 Mutation Contributes to Oncogenic Reprogramming through Aberrant Histone Modification and Localization within the Nucleus" (2026). Alpha Omega Alpha Research Symposium Posters. 17.
https://jdc.jefferson.edu/aoa_research_symposium_posters/17



Comments
Presented at the 2026 AOA Research Symposium.